 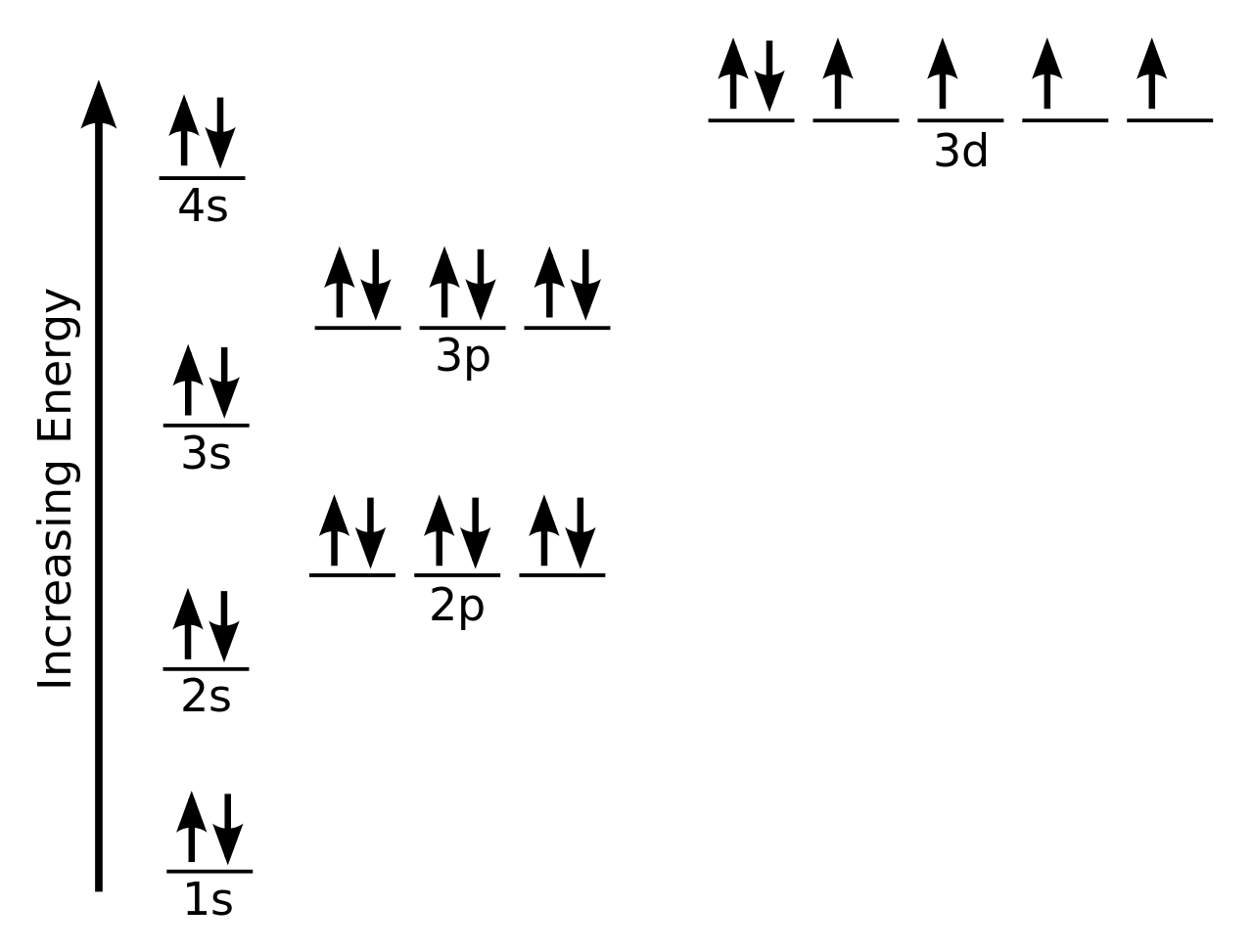
For example, the abbreviated configuration for Neon is 2s 2 2p 6. In the periodic table beyond Helium (He), each element's electron configuration is shown in an abbreviated form that starts with the symbol of the noble gas that precedes it. The superscript shows that there is one electron in the 1s orbital. The simplest configuration is for Hydrogen: 1s 1. Also, I'm skeptical about whether you can measure a state of $p_xp_z$ given that $p_x$ and $p_z$ are eigenstates of non-commuting operators, but let's leave this aside for now (or assume, perhaps, that we're dealing with $p_z$).An atom's electron configuration describes the distribution of its electrons in the atomic orbitals ordered by the orbitals' energy levels. * You can't measure individual electrons in an atom, you can only measure their collective properties. In a molecule, electrons don't remember "where" they come from, so it becomes pointless to quibble over whether it was in the $p_x$ or $p_y$ orbital. Also, it's perhaps not worth overthinking this: once you form a bond, all of this ceases to have any meaning. The answer is that it's far easier than dealing with a superposition of states (in (3)), and since space is isotropic, $xy$ is the same as $xz$ and $yz$ as long as you relabel the axes, which means that (2) is more of a technicality rather than a real "gotcha". Having gone through this, there is still the question of why we write $p_xp_y$ when it's not really accurate. My old question: What constraints are imposed on a wavefunction by the symmetry of the system? is somewhat related, except that it deals with boron (which has the same issue). It's better to posit that an isolated carbon atom is in a superposition of those three states, i.e. The previous point assumes that you have made a measurement of the electrons, such that you can definitively say that they are in a specific set of orbitals.* But prior to this measurement, there is no reason that they should already be in that state. Again, this symmetry stems from the fact that there is no physical factor that favours any of the three coordinate axes over each other. Instead, on average, you would have one-third of them with the configuration $(p_x)^1(p_y)^1$, one-third with $p_xp_z$, and one-third with $p_yp_z$. If you kept these coordinate axes consistent between different atoms, such that every $p_x$ orbital pointed in the same direction, you should not expect to find in all molecules that the $p_x$ and $p_y$ orbitals are always occupied. You can still arbitrarily assign $x$-, $y$-, and $z$-labels to three perpendicular axes. There is nothing that can physically differentiate the $x$-axis from the $y$-axis or the $z$-axis. More formally, this reflects the isotropy of the system, which means that all directions are equal. That is to say, the $p_x$, $p_y$, and $p_z$ orbitals are all interchangeable. I'll try and give a short summary: each point goes slightly deeper than the previous one.įor an isolated atom, the labelling of the orbitals is arbitrary. The answer comes in several layers, as Martin has alluded to. Why is this the case, and not, for example, 1s 2 2s 2 2p y 1 2p z 1? Update: I found this question and this answer which both state that non-hybridized carbon has the configuration 1s 2 2s 2 2p x 1 2p y 1.  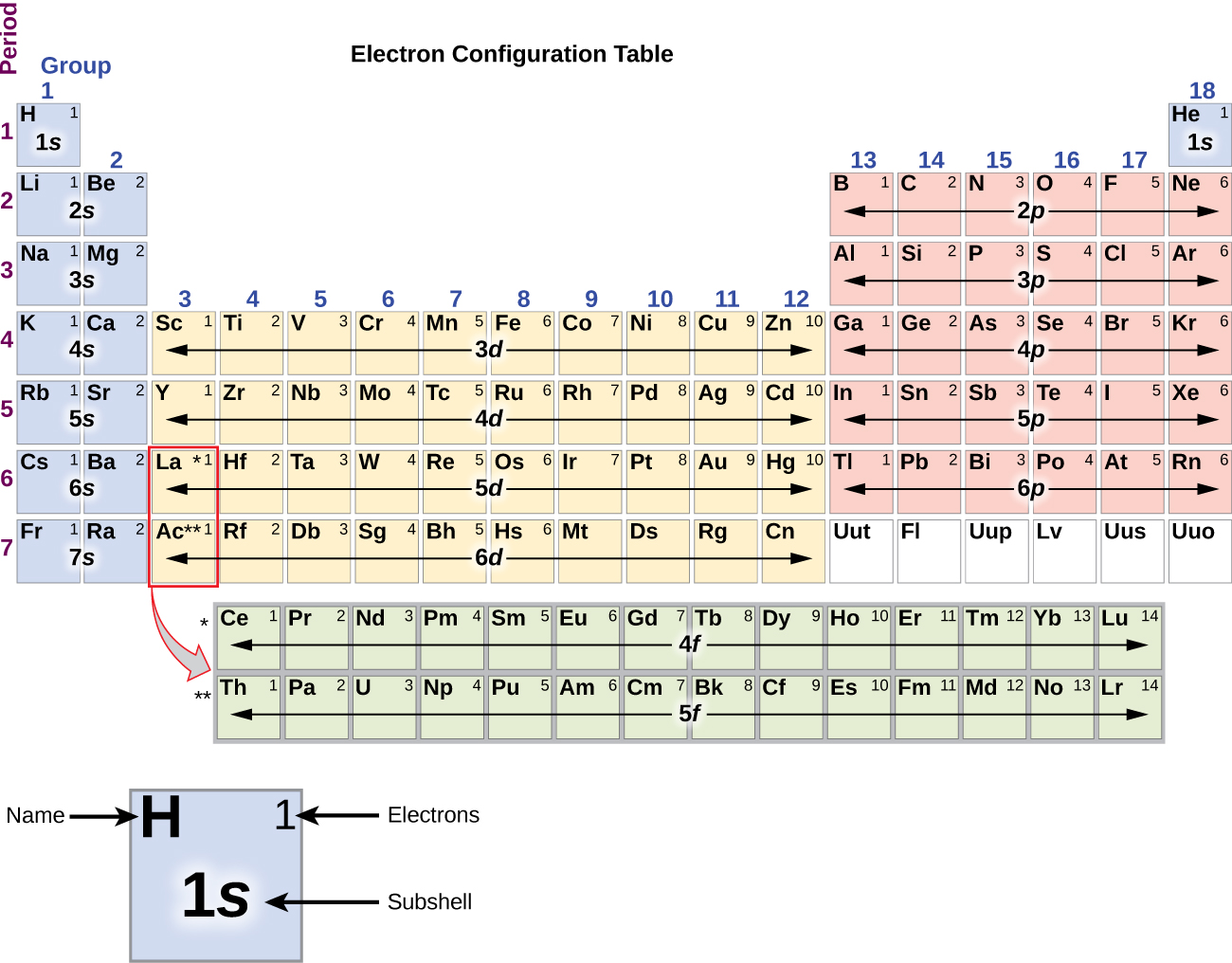
However, how are the second shell electrons distributed in a non-hybridized carbon atom? Does such an atom even naturally occur or does it always hybridize? It then makes sense that the four electrons available each occupy one of the four new orbitals (so that each of them holds one electron), because this way, they can have the greatest spatial distance to each other (I assume they "want" this as they repel each other). I also learned that the three p orbitals can be combined with the 2s orbital (sp 3 hybridization) to form four new hybrid orbitals. Which orbitals do they occupy? Since their energy levels are identical, is it random? However, there are only two electrons to be distributed on these three orbitals. However, there are actually three p-orbitals in the second shell, namely p x, p y and p z, which could each hold two electrons. I see that carbon has an electron configuration of 1s 2 2s 2 2p 2. 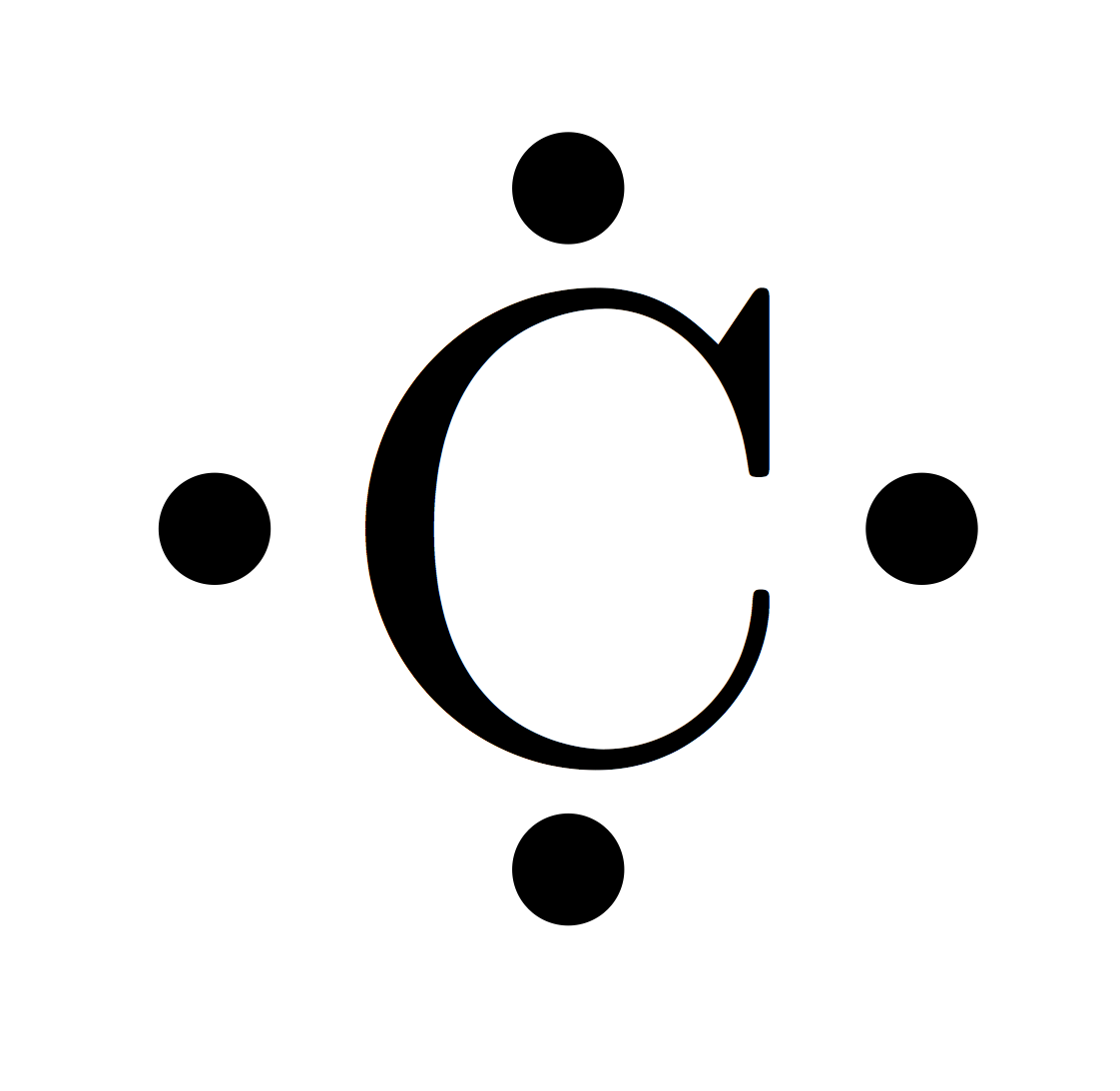
I'm currently learning about orbital hybridization in carbon. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
